Meningeal lymphatics clear erythrocytes that arise from subarachnoid hemorrhage
Meningeal lymphatics clear erythrocytes that arise from subarachnoid hemorrhage"
- Select a language for the TTS:
- UK English Female
- UK English Male
- US English Female
- US English Male
- Australian Female
- Australian Male
- Language selected: (auto detect) - EN
Play all audios:
ABSTRACT Extravasated erythrocytes in cerebrospinal fluid (CSF) critically contribute to the pathogenesis of subarachnoid hemorrhage (SAH). Meningeal lymphatics have been reported to drain
macromolecules and immune cells from CSF into cervical lymph nodes (CLNs). However, whether meningeal lymphatics are involved in clearing extravasated erythrocytes in CSF after SAH remains
unclear. Here we show that a markedly higher number of erythrocytes are accumulated in the lymphatics of CLNs and meningeal lymphatics after SAH. When the meningeal lymphatics are depleted
in a mouse model of SAH, the degree of erythrocyte aggregation in CLNs is significantly lower, while the associated neuroinflammation and the neurologic deficits are dramatically
exacerbated. In addition, during SAH lymph flow is increased but without significant lymphangiogenesis and lymphangiectasia. Taken together, this work demonstrates that the meningeal
lymphatics drain extravasated erythrocytes from CSF into CLNs after SAH, while suggesting that modulating this draining may offer therapeutic approaches to alleviate SAH severity. SIMILAR
CONTENT BEING VIEWED BY OTHERS MENINGEAL LYMPHATIC VESSELS MEDIATE NEUROTROPIC VIRAL DRAINAGE FROM THE CENTRAL NERVOUS SYSTEM Article 06 May 2022 MENINGEAL LYMPHATIC DRAINAGE: NOVEL INSIGHTS
INTO CENTRAL NERVOUS SYSTEM DISEASE Article Open access 05 May 2025 PRELIMINARY RESULTS IN THE ANALYSIS OF THE IMMUNE RESPONSE AFTER ANEURYSMAL SUBARACHNOID HEMORRHAGE Article Open access
16 July 2020 INTRODUCTION As estimated by Global Burden of Diseases (GBD 2016), stroke is the second leading cause of death1. Subarachnoid hemorrhage (SAH) contributes to only 5% of the
cases of stroke2, but it occurs at a fairly young age and is associated with a high degree of mortality (in the range of 50%)3. Thus SAH is a large burden on society. The progression of SAH
includes early brain injury that appears the first 3 days after injury, followed by delayed cerebral ischemia (DCI) 3–4 days after injury, which reaches the highest incidence and severity
6–8 days after SAH4. As vasospasm, microthrombosis, and neuroinflammation contribute greatly to the DCI and clinical prognosis, pharmacological approaches to treat this pathology have been
focused on preventing vasospasm, microclot formation, and anti-inflammation for decades; however, to date, few agents have been found to exert beneficial effects on patient outcomes5,6. The
pathologies of SAH are caused by the extravasated blood presenting in the subarachnoid space (SAS). Thus a better understanding of extravasated blood clearance may help in the development of
effective therapeutic approaches. When a SAH attack occurs, blood releases into the SAS, leading immediately to clot formation, which disappear within 2–3 days7,8. To date, clot lysis and
phagocytosis by macrophages and neutrophils were considered as ways to clear the extravasated blood and the subsequent clots in the SAS9,10. However, the mechanism(s) by which extravasated
blood is cleared is still unclear. Recently, lymphatic vessels have been rediscovered and characterized in the meninges surrounding the central nervous system11,12. These lymphatics are
responsible for the drainage of cerebrospinal fluid (CSF) macromolecules and immune cells to the cervical lymph nodes (CLNs)13,14. Meningeal lymphatics aid in the clearance of amyloid-beta
in aged mice and transgenic mouse models of Alzheimer’s disease, and the augmentation of meningeal lymphatic function alleviates age-associated cognitive impairment13,15,16,17. But in an
experimental autoimmune encephalomyelitis (EAE) mouse model the ablation of meningeal lymphatics or inhibition of vascular endothelial growth factor receptor 3 (VEGFR3) diminished the EAE
severity and the inflammatory response of brain-reactive T cells14,18. Thus the meningeal lymphatics may play different roles in different neurological diseases. Embryonic mesenteric
lymphatic vessels have recently been shown to clear extravascular red blood cells (RBCs) leaking from the adjacent developmental vascular remodeling, suggesting that the lymphatic system has
the potential to clear the erythrocytes in interstitial fluid19. But, as noted above, while meningeal lymphatics drain immune cells and macromolecules from CSF, it is still unclear whether
they clear the extravasated blood after SAH. Here we report that the extravasated blood is aggregated in CLNs, and the erythrocytes are accumulated in the lymphatics of CLNs inside and
around meningeal lymphatic vessels for 4 h after SAH. We use CFSE (5-(and 6)-carboxyfluorescein diacetate succinimidyl ester) to label erythrocytes ex vivo and inject them into the cisterna
magna and demonstrate that the labeled erythrocytes are also evident in the lymphatics of CLNs and meningeal lymphatics. When we ablate meningeal lymphatics by photoconverted visudyne, RBC
drainage into CLNs is significantly reduced, while the neuroinflammation and the neurological deficits associated with SAH are exacerbated. We further block VEGFR3 and also observe a worse
brain injury during SAH. We examine the structure and functional characteristics of meningeal lymphatics during neuroinflammation and find the lymph flow is augmented but lymphangiogenesis
and expansion are not pronounced. Our findings demonstrate that meningeal lymphatics participate in the drainage of RBCs into CLNs during the very early stage of SAH and may be a potent
target in the treatment of this pathology. RESULTS RBCS IN CSF ARE DRAINED BY THE MENINGEAL LYMPHATICS TO CLNS Tracers, proteins, and labeled T cells injected into the brain or CSF are found
in CLNs, indicating the drainage function by meningeal lymphatics14,20. To explore whether the erythrocytes released into the CSF, a condition called SAH, are drained to CLNs via the
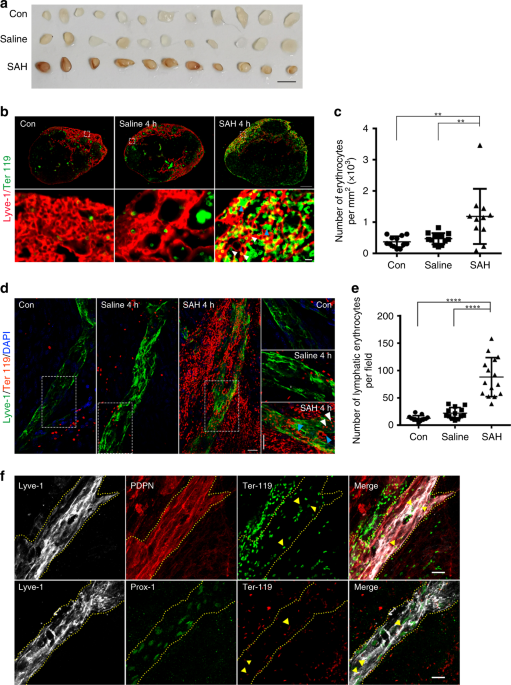
meningeal lymphatics, we injected autologous blood into the cisterna magna of mouse. Four hours later, we observed that the deep CLNs (dCLNs) and mandibular LNs were infused with blood,
while no evidence of blood was observed in the saline-injected and control groups (Fig. 1a, Supplementary Fig. 1a, b). The data also suggest that the superficial parotid LNs, though located
in the superficial anterior neck, did not drain the CSF erythrocytes, and there is no drainage of the axillary, the brachial, and the tracheobronchal LNs (Supplementary Fig. 1a). Next, we
used anti-Ter119 antibody to label erythrocytes and anti-Lyve-1 antibody to visualize the lymphatics of CLNs. The number of Ter119-positive erythrocytes in the Lyve-1-positive lymphatic
sinus was significantly greater in the SAH group when compared with the saline and control groups for both types of CLNs (for the comparison of dCLNs, _P_ = 0.002, Con vs SAH, _P_ = 0.0063,
saline vs SAH, Fig. 1b, c; Supplementary Fig. 1c, d). We found that the Ter119-positive cells were distributed mainly in the Lyve-1-positive lymphatic sinus of CLNs. To determine whether the
erythrocytes are drained to CLNs via the meningeal lymphatics, we isolated the meninges and stained the lymphatic vessels with antibodies. Erythrocytes were observed to accumulate around
and inside of meningeal lymphatics at 4 h post-induction of SAH, while erythrocytes were not seen significantly entering into the lymphatics of the control and saline-injected groups (_P_
< 0.0001, Con vs SAH; _P_ < 0.0001, saline vs SAH; Fig. 1d, e). The corresponding orthogonal view (Supplementary Fig. 2) showed the erythrocytes co-localizing with Lyve-1-positive
lymphatic endothelial cells, suggesting that these cells were drained via the meningeal lymphatic vessels. The identification of meningeal lymphatics was further confirmed by co-labeling
them with other classical lymphatic endothelial cell markers including podoplanin (PDPN) and Prox1 with lyve-1, and the erythrocytes also showed accumulation into meningeal lymphatics (Fig.
1f). Morphologically intact erythrocytes and clusters of degraded membranes were seen at the same time in the lymphatics of CLNs and meninges (Fig. 1b, d), though it is not clear whether the
erythrocytes were broken down before transportation by the lymphatics or after. But the data suggest that at least some erythrocytes were drained into CLNs before degradation. To further
confirm that the erythrocytes in meningeal lymphatics and CLNs were drained from exogenous injection, we labeled erythrocytes with CFSE in vitro and then injected them into the cisterna
magna. Four hours later, the labeled erythrocytes also showed similar accumulation in the meningeal lymphatics (_P_ < 0.0001, Fig. 2a, b), dCLNs (_P_ = 0.0006, Fig. 2c, e), and mandibular
LNs (_P_ = 0.0207, Fig. 2d, e), while the saline-treated group did not show any evidence of CFSE-labeled erythrocytes. ABLATIVE MENINGEAL LYMPHATICS BLOCK RBC DRAINAGE TO CLNS To further
verify whether meningeal lymphatic vessels serve as the route for the drainage of erythrocytes from the CSF into the CLNs, we ablated meningeal lymphatic vessels by injecting visudyne into
the cisterna magna and photoconverted it by laser light. After 7 days of ablation, lymphatic coverage of the transverse sinus (_P_ < 0.0001, Con vs Laser + Visudyne; _P_ < 0.0001,
Laser vs Laser + Visudyne; _P_ < 0.0001, Visudyne vs Laser + Visudyne; Fig. 3a, b) and the superior sagittal sinus were notably lower (_P_ < 0.0001, Con vs Laser+Visudyne; _P_ =
0.0007, Laser vs Laser + Visudyne; _P_ = 0.0003, Visudyne vs Laser + Visudyne; Fig. 3c). No difference in cerebral blood flow was observed between control mice and the Laser + Visudyne group
(Supplementary Fig. 3a, b). The coverage of blood vasculature on the transverse sinus and superior sagittal sinus were not altered in the Visudyne-photoconverted group compared with Laser
only or Visudyne only group (Supplementary Fig. 3c, d). On the seventh day after the ablation of the meningeal lymphatics, we induced SAH. After 4 h of SAH, blood infusion into the dCLNs and
mandibular LNs were significantly lower in the mice with impaired meningeal lymphatics compared to the Laser only and the Visudyne only groups (Fig. 3d, Supplementary Fig. 3e, f). And the
number of Ter119-labeled erythrocytes was markedly lower in the dCLNs (_P_ = 0.0013, L + SAH vs L + V + SAH; _P_ = 0.0060, V + SAH vs L + V + SAH; Fig. 3e, f) and mandibular LNs
(Supplementary Fig. 3g, h) of the lymphatic-ablation group. In general, blood is released into the SAS when SAH occurs and is cleared within 2–3 days7,8. But we found that clot clearance was
blocked when the meningeal lymphatics were ablated as clots persisted in the brain until 7 days after the induction of SAH in the Laser + Visudyne group (Fig. 3g). ABLATION OF MENINGEAL
LYMPHATICS WORSENS SAH SEVERITY Neuroinflammation is prominent in SAH, leading to cerebral cell damage and vasospasm21. Microglia are resident brain macrophages, can be activated in SAH, and
can display either classical pro-inflammatory phenotype or alternative anti-inflammatory phenotype polarization22. We hypothesized that, after ablation of meningeal lymphatics, the
microglia activation would be worsened owing to the prolonged exposure to erythrocytes degradants. We thus performed whole-brain flow cytometry to determine the ratio of CD16/32-posititive
pro-inflammatory microglia and CD206-positive anti-inflammatory microglia. For these measurements, CD11b+CD45low cell populations were gated as myeloid lineage cells including microglia, in
which the CD16/32 was used as a marker for cells with pro-inflammatory phenotype and CD206 was used as a marker for cells with anti-inflammatory phenotype. The gating strategy is shown in
Fig. 4a. Compared with SAH mice with intact lymphatics (SAH only, L + SAH and V + SAH group), SAH mice with lymphatic ablation (L + V + SAH group) showed a significantly greater percentage
of CD16/32+CD206− microglia (_P_ < 0.0001 SAH/L + SAH/V + SAH vs L + V + SAH, Fig. 4b, c) and a lower percentage of CD206+CD16/32− microglia (_P_ = 0.0146, SAH vs L + V + SAH; _P_ <
0.0001, L + SAH vs L + V + SAH Fig. 4b, d). When compared with the Laser + Visudyne group, the degree of activated microglia polarized to a pro-inflammatory phenotype was markedly greater
after SAH as evidenced by an increased ratio of CD16/32+CD206− subsets and decreased ratio of CD206+CD16/32− cells. (_P_ < 0.0001, L + V vs L + V + SAH for CD16/32+CD206− and _P_ =
0.0086, L + V vs L + V + SAH for CD206+CD16/32−, Fig. 4c, d). The percentages of CD16/32 and CD206 double-positive cells, an intermediate state of polarization, and difference were only
observed in the comparison between V + SAH and L + V + SAH groups (_P_ = 0.025, Fig. 4e). To assess the impact of the ablation of meningeal lymphatics on neurological function of the mice
with SAH, we applied the open field test to evaluate the exploratory behavior of mice. SAH mice in the lymphatic-ablated group showed a significant decrease in the percentage of time spent
in the center and the number of entrances into the center compared to the non-ablated SAH mice (_P_ = 0.0194, SAH vs L + V + SAH, _P_ = 0.0053, L + SAH vs L + V + SAH; _P_ = 0.009, V + SAH
vs L + V + SAH, Fig. 4f, _P_ < 0.0001, SAH vs L + V + SAH; _P_ = 0.0001, L + SAH vs L + V + SAH; _P_ = 0.0018, V + SAH vs L + V + SAH; Fig. 4g). We also used a Y-maze test to evaluate the
short-term working memory of the SAH model. We found that the SAH mice with ablated meningeal lymphatics were more vulnerable than the mice with intact lymphatics, as demonstrated by the
percentage of time spent in the novel arm (NA) (_P_ = 0.0006, SAH vs L + V + SAH; _P_ = 0.003, L + SAH vs L + V + SAH, Fig. 4h) and the number of entrances into the NA (_P_ < 0.0001, SAH
vs L + V + SAH; _P_ < 0.0001, L + SAH vs L + V + SAH; _P_ = 0.0338, V + SAH vs L + V + SAH, Fig. 4i). When compared with the mice only treated with photoconverted visudyne, the mice
accompanied with SAH showed worse performance in both the open field (_P_ = 0.0138, time spent in the center; _P_ = 0.0141, number of entries into the center; L + V vs L + V + SAH, Fig. 4f,
g) and the Y-maze test (_P_ = 0.0198, time spent in the NA arm; _P_ = 0.0303, number of entries into the NA arm, Fig. 4h, i). INHIBITION OF VEGFR3 EXACERBATES SAH PATHOLOGY Meningeal
lymphatics maintain the potential to grow or regress in adults13,18. To further confirm the lack of integrity of meningeal lymphatics resulting in unfavorable outcomes in SAH, we inhibited
VEGFR3, a tyrosine kinase receptor that promotes lymphangiogenesis, with MAZ51, a chemical inhibitor of VEGFR3 with proven effectiveness in inhibiting lymphangiogenesis18. MAZ51 was given
intraperitoneally for 30 days (Fig. 5a). As shown in Fig. 5b, meningeal lymphatics underwent regression after treatment with MAZ51, as evidenced by a reduction in the lyve-1-positive area in
both the transverse and superior sagittal sinuses (Fig. 5c). We observed no change in cerebral blood flow (Supplementary Fig. 3a, b) and of CD31-positive blood vasculature area of the
sinuses (Supplementary Fig. 3i–j). On the 30th day after treatment, vehicle- and MAZ51-treated mice were injected autologous blood or saline. Microglia activation was also detected by flow
cytometry (the general gating strategy is shown in Fig. 5d). As our data show, the percentage of CD16/32+CD206− pro-inflammatory microglia was increased after SAH induction (_P_ = 0.0139,
Vehicle + Sham vs Vehicle + SAH; _P_ < 0.0001, MAZ51 + Sham vs MAZ51 + SAH, Fig. 5e, f), but the proportion of activated microglia polarized to CD16/32+CD206− phenotype were greater in
the mice with meningeal lymphatic regression (_P_ < 0.0001, Vehicle + SAH vs MAZ51 + SAH, Fig. 5f). The percentage of CD206+CD16/32− anti-inflammatory microglia was not markedly affected
by SAH or MAZ51 induction but significantly reduced after SAH + MAZ51 treatment (_P_ = 0.0233, MAZ51 + SAH vs Vehicle + Sham) (Fig. 5g). For the cell population characterized by
CD16/32+CD206+, no statistical significance was observed (Fig. 5h). These results were consistent with the above findings, indicating that the deterioration of the meningeal lymphatic
vessels leads to further aggravation of neuroinflammation caused by SAH. To validate the regression of meningeal lymphatics resulting in worse neurological outcomes after SAH, we performed
the behavioral tests on MAZ51-induced lymphatic regression mice. The exploratory behavior and short-term working memory were assessed by open field test and Y-maze test, respectively. Mice
with degenerated meningeal lymphatics spent less time in the center after SAH (_P_ = 0.0111, Vehicle + SAH vs MAZ51 + SAH, Fig. 5i) but did not show any difference in the number of entries
into the center (Fig. 5j). The deficits of short-term working memory after SAH also worsened in mice treated with MAZ51, represented by less time spent in the NA (_P_ = 0.0001, MAZ51 + Sham
vs MAZ51 + SAH, Fig. 5k) and the number of entries into the NA (_P_ = 0.0037, MAZ51 + Sham vs MAZ51 + SAH; _P_ = 0.0221, Vehicle + SAH vs MAZ51 + SAH, Fig. 5l). CHARACTERIZATION OF MENINGEAL
LYMPHATICS IN SAH It has been reported that during acute inflammation, such as in intestinal inflammation, colitis, endocarditis, and rheumatoid arthritis, lymphatic flow and
lymphangiogenesis are increased in the local peripheral lymphatic vessels, which is a mechanism for the body to reduce inflammation and edema23,24,25,26,27,28. To explore whether the
neuroinflammation following SAH affects the function of the meningeal lymphatic vessels, we injected AF488-conjugated anti-Lyve-1 or fluorescent microbeads into the cisterna magna at day 7
after induction of SAH or saline injection. After 30 min, meninges were harvested and stained for Lyve-1 using AF555-conjugated secondary antibody. The percentage of meningeal lymphatics
labeled by AF488-anti-Lyve-1 antibody (intracisterna magna (i.c.m.)) indicates the speed of meningeal lymphatic flow. A significantly greater percentage of meningeal lymphatics labeled by
AF488 anti-Lyve-1 (i.c.m.) was seen in the SAH group vs the controls (_P_ = 0.0122, Con vs SAH; _P_ = 0.0303, Saline vs SAH; Fig. 6a–c), and all the lymphatics labeled by injected antibody
were seen on the transverse sinus. A greater percentage of lymphatics labeled by AF488 anti-Lyve-1 (i.c.m.) also could be seen in dCLNs in the SAH group vs controls (_P_ = 0.0183, Con vs
SAH; _P_ = 0.0181, saline vs SAH; Fig. 6d, e). Similarly, 2 h after fluorescent microbead injection, the bead coverage in dCLNs was significantly higher in the SAH group vs the controls (_P_
= 0.0004, Con vs SAH; _P_ = 0.0251, saline vs SAH; Fig. 6f–h). In addition, the higher contraction frequency of mandibular afferent lymphatic vessels was detected in the SAH group than that
in the saline group (_P_ = 0.0261, Fig. 6i, j). To investigate whether the SAH process affects growth and expansion of the meningeal lymphatic vessels, we divided the meningeal lymphatics
on the transverse sinus into eight different segments and measured the diameter and branching of each segment. Although we found significant dilation of vessel diameter (Fig. 7a, b) and a
greater number of branches (Fig. 7a, c) in some segments in the SAH group, the lymphatic vessel area in the transverse sinus and the superior sagittal sinus did not change (Fig. 7d, e). Thus
we conclude that lymphangiogenesis and lymphangiectasia of the meningeal lymphatics did not significantly alter at 7 days post-induction of SAH. DISCUSSION SAH most commonly occurs due to
the rupture of an aneurysm in the cerebral artery, releasing blood into the SAS and resulting in a series of early neurological complications and DCI. Understanding the process by which
meningeal lymphatics drain fluid, macromolecules and immune cells from the CSF has shed a light on the pathogenesis of neurological diseases, including Alzheimer’s disease, multiple
sclerosis, and ischemic brain injury in past 5 years13,14,16,18,29,30. However, whether the extravasated erythrocytes released into the CSF during SAH can be removed by meningeal lymphatics
remains unclear. Here we show that the extravasated erythrocytes in the SAS are drained into dCLNs and mandibular LNs through the meningeal lymphatics and that the depletion of meningeal
lymphatics blocks the clearance of extravasated blood. During SAH, blood pours into the SAS, where erythrocytes break down and release hemoglobin (Hgb) and its products that contribute to
brain injury31,32. Previous studies report that extravasated erythrocytes and their degradation products in the SAS can be cleared via mechanisms of clot lysis or phagocytosis7,9,10,33. With
the upregulation of intercellular adhesion molecule-1 in cerebral blood vessel endothelial cells, macrophages and neutrophils enter the SAS and then phagocytose extravasated erythrocytes
and Hgb34,35,36. It is proposed that macrophages and neutrophils are trapped in the SAS after phagocytosis of extravasated erythrocytes and Hgb, which then die and are degranulated within
2–4 days33,35. However, the structure and function of meningeal lymphatics have been defined recently11,12. Furthermore, microglia also play a role to clear in SAH by expressing heme
oxygenase-137. Microglial activation and monocyte infiltration are observed 24 and 72 h after SAH, respectively38. Here we show that erythrocytes can be detected in the CLNs (dCLNs and
mandibular LNs) and meningeal lymphatics at 4 h post SAH, a time much early than macrophage/microglia-mediated clearance. These findings reveal that the extravasated erythrocytes can be
drained into CLNs before being degraded into Hgb or phagocytosed by macrophages and neutrophils, at least in the very early stages of SAH (4 h after SAH in this study). Meanwhile, we found
here that the depletion of meningeal lymphatics significantly blocked the drainage of extravasated erythrocytes into CLNs, demonstrating that meningeal lymphatics serve as the route of
draining for erythrocytes into the CLNs. The blood clots were observed in the brain of SAH mice with ablated lymphatics indicating that the clot clearance may be affected by the reduced
cerebral lymphatic drainage. As CSF flow is not unidirectional, ventricular blood presenting in some acute SAH is proposed that is refluxed from cisternal hemorrhage and not indicative of
primary ventricle bleeding39,40. Thus we considered that the clots on the pons and medulla in this study may result from the ventricular blood refluxed from SAS. There are several potential
routes of extravasated blood clearance as discussed above. Different routes may participate in different phases of SAH, for example, before and after the degradation of erythrocytes. Great
effort should be made in the future to clarify the kinetics and relative contribution of each of these pathways of extravasated erythrocyte clearance. This study reveals a lymphatic route
for clearing extravasated erythrocytes in SAH; however, the mechanism by which extravasated erythrocytes enter the meningeal lymphatics is still unclear. T cells and dendritic cells enter
into and migrate through the meningeal lymphatics and peripheral lymphatic system via the C-C chemokine motif receptor 7–C-C chemokine motif ligand 21 pathway14,18,41,42. Macromolecules can
be endocytosed by brain lymphatic endothelial cells in zebrafish43. Fluids and solutes diffuse into the peripheral lymphatic lumen due to the pressure difference between that of the
interstitial fluid and the lumen44, while chylomicron uptake into lacteals occurs by active transport vesicles through lymphatic endothelial cells45,46. Little is known about the mechanism
of extravasated erythrocytes entering meningeal lymphatics, but the known ways by which immune cells and macromolecules enter into the lymphatic lumen may provide some hints for future
research. Neuroinflammation that occurs as a result of SAH is caused by the accumulation of erythrocyte degradation products, including Hgb, methemoglobin, heme, and hemin in the SAS.
Microglia are activated and accumulated in the brain accompanied by upregulation of inflammatory cytokines, including tumor necrosis factor-α, interleukin (IL)-1β, and IL-6. These
inflammatory factors contribute to the neuronal cell death and secondary brain injury after SAH38,47,48,49. In this study, we found that microglial activation and polarization into a
pro-inflammatory microglial cells in SAH are aggravated by the depletion of meningeal lymphatics. This exacerbation may have occurred because the extravasated erythrocytes were trapped in
the SAS, thus increasing the accumulation of Hgb and its products and prolonging brain exposure to these degradation products. A previous study reported that the subarachnoid clot volume and
spontaneous clearance rate are closely related to vasospasm8. Thus promoting the clearance of subarachnoid erythrocytes via accelerating meningeal lymphatic flow may be a potential therapy
for the pathologies associated with SAH. The enhancement of lymphatic draining function, lymphangiogenesis, and lymphangiectasia are commonly observed in peripheral acute inflammation,
including arthritis, bacterial keratitis, and colitis50,51,52, with the enhanced lymphatic flow and lymphangiogenesis reducing the local inflammation and edema. The overexpression of VEGF-C
by viral infection or local injection has been shown to ameliorate, while the blockade of the VEGF-C/VEGFR-3 pathway exacerbates, inflammation52,53,54,55,56. Increasing meningeal lymphatic
drainage was observed at day 7 of SAH in this study; however, lymphatic expansion and growth were not pronounced. These results are in consistent with previous reports that the morphology of
meningeal lymphatics did not change in EAE-associated neuroinflammation14,18. Meningeal lymphatics may have a limited growth capability when exposed to neuroinflammation. Further research
is needed to determine whether modulating lymphangiogenesis by overexpression of VEGF-C or other means affects SAH induced-neuroinflammation. In summary, we show that extravasated
erythrocytes in the SAS are drained into CLNs through meningeal lymphatics during SAH. This study adds insight into the extravasated erythrocyte clearance pathway that occurs during the very
early stages of SAH and provides a possible therapeutic avenue for its treatment, as well as possibly other types of intracranial hemorrhage. MATERIALS AND METHODS ANIMALS Specific
pathogen-free, C57BL/6 male mice (6–8 weeks old) were purchased from Shanghai Model Organisms Center. Mice were housed in the animal facility with controlled habituation and temperature, on
12-h light vs dark cycles, and fed with regular rodent’s chow and sterilized tap water ad libitum. Mice were allowed to accommodate for 2 weeks before experimental procedures. All animal
procedures were approved by Longhua Hospital - Animal Ethics Committee and were performed according to the Guiding Principles for the Care and Use of Laboratory Animals Approved by Animal
Regulations of National Science and Technology Committee of China. INDUCTION OF SAH An SAH model was established according to a previous publication57. Briefly, 60 µl autologous blood was
withdrawn from the right femoral artery after mice were anesthetized with ketamine hydrochloride (100 mg/kg, Fujian Gutian Pharma Co., Ltd, 1505223). The animal’s head was fixed in a
stereotactic frame (RWD), the posterior neck was incised, the posterior neck muscles were separated to access the cisterna magna, and 60 µl of autologous blood was injected at a low rate
into this region. The needle was kept in place for 2 min to prevent backflow or CSF leakage. Sham-treated mice were similarly injected with 60 µl of saline. Then the mice were sutured and
kept on a 37 °C heating pad (Thermo Plate) until entirely recovered from anesthetization. Mice in blank control group (Con group) were housed as usual and did not receive any additional
treatment. BEHAVIORAL ANALYSIS The open field test was used to evaluate spontaneous activity and exploration behaviors. Mice moved freely in the box (60 cm × 60 cm × 25 cm) for 10 min, and
then the distance traveled, the time spent in the center, and the number of entrances into the center area was recorded using the videotracking software EthoVision XT 12 (Noldus). Short-term
memory was assessed by Y-maze test. The maze included the starting arm, the NA, and the other arm. Before the test, mice underwent a 5-min training period with a block of the NA in the
maze. Two hours later, the NA was opened, and the mice were allowed to travel freely throughout the three arms, with the percentage of time spent in the NA and the number of entries into the
NA in 5 min recorded by ANY-maze (Stoelting, America). I.C.M. INJECTION Mice were fixed in a stereotactic frame (RWD) after anesthetization with ketamine hydrochloride, and an incision was
done along with separation of the posterior neck muscles to access the cisterna magna. Then 2 µl of fluorescent microbeads (Latex beads, amine-modified polystyrene, fluorescent red, Cat. No.
L2778-1, Sigma) were injected into the cisterna magna at a rate of 0.5 µl/min or 5 µl of Alexa Fluor 488-conjugated anti-Lyve-1 antibody (AF488 anti-Lyve-1) (Cat. No. 53-0443-82,
eBioscience) was injected into the cisterna magna at a speed of 1 µl/min. After injection, the needle was left in place for 2 min to prevent backflow and leakage. Then the mice were sutured
and kept on a 37 °C heating pad until responsive. The AF488 Lyve-1 antibody was left to flow for 30 min and the fluorescent microbeads were left to flow for 2 h before the mice were killed.
VISUDYNE TREATMENT To ablate the meningeal lymphatics, visudyne treatment was carried out according to a previous publication14. Briefly, mice were anesthetized with ketamine hydrochloride
and their heads were fixed in a stereotactic instrument. Visudyne (APExBIO, Cat. No. A8327) was reconstituted according to the manufacturer’s instructions, and 5 µl was injected into the
cisterna magna at a speed of 1 µl/min. Fifteen minutes later, a nonthermal 689-nm wavelength laser light (Changchun Laser Technology), with a dose of 50 J/cm2 and intensity of 600 mW/cm2,
was applied on 5 different spots through the skull (the injection site, left and right transverse sinuses, the superior sagittal sinus, and the junction of all sinuses). For the Laser group,
mice underwent the same procedures of laser treatment but without visudyne injection, and for the Visudyne group, mice were just given 5 µl of visudyne into the cisterna magna without laser
treatment. During the laser procedure, the eyes of the mice were protected. Then the incision in the mice was sutured and the mice were kept on a 37 °C heating pad until entirely recovered
from anesthetization. VEGFR3 TYROSINE KINASE INHIBITOR ADMINISTRATION MAZ51 (Cat. No. 676492, Merck Millipore) was dissolved in dimethyl sulfoxide and intraperitoneally injected at 10 mg/kg
of body weight for 30 days (5 days per week). The control group was given the same volume of vehicle. On the 30th day, mice of both groups were divided into either an autologous blood
injection or a saline injection group. On the seventh day after SAH induction, mice were killed for analysis. ERYTHROCYTE ISOLATION AND LABELING Whole blood was collected from mice from the
right femoral artery after they were anesthetized, then 1:1 diluted with 2% fetal bovine serum (FBS)–phosphate-buffered saline (PBS), followed by centrifugation for 10 min (800 × _g_)
without braking. The plasma and buffy coat layers were removed, the erythrocytes were collected from the bottom of tubes, and the cells were diluted into 106 cells/ml, then CFSE (20 μM/ml,
eBioscience, Cat. No. 65-0850-84) was added, followed by incubation for 10 min in 37 °C. After washing with 2% FBS–PBS, erythrocytes (about 106 cells in 60 µl) were injected into the
cisterna magma. The control group mice were injected with saline. Four hours after erythrocyte was injected, meninges, dCLNs, and mandibular LNs were harvested. FLOW CYTOMETRY Mice brains
were dissected after transcardial perfusion by cold PBS, then minced into small pieces. Brain tissue was digested by collagenase A (1 mg/ml, Sigma Aldrich, Cat. No. 10103578001) for 30 min
at 37 °C, then filtered by 70-μm nylon mesh cell strainers (BD bioscience). A cell suspension was made with 30% stock isotonic percoll (SIP) (GE, 17089109) and layered on the top of 70% SIP
and then centrifuged at 500 × _g_ at 25 °C for 30 min without braking. Cells were collected from the 70–30% SIP interphase and stained for live cells by Fixable Viability Dye eFluorTM 780
(Cat. No. 65-0865-18, eBioscience), extracellular markers with the following antibodies at a 1:100 dilution: rat anti-CD11b fluorescein isothiocyanate (FITC)-conjugated antibody (11-0112-82,
eBioscience), rat anti-CD45 PerCP-Cy5.5-conjugated antibody (45-0451-82, eBioscience), rat anti-CD16/32 allophycocyanin (APC)-conjugated antibody (558636, BD Bioscience) and intracellular
marker rat anti-CD206 R-phycoerythrin (PE)-conjugated antibody (12-2061-80, eBioscience). The corresponding isotype control antibodies that were used are as follows: Rat IgG2b κ Isotype
control FITC-conjugated antibody (11-4031-82, eBioscience) Rat IgG2a κ Isotype control PerCP-Cy5.5-conjugated antibody (45-4321-80, eBioscience), Rat IgG2b κ Isotype control PE-conjugated
antibody (12-4031-82, eBioscience), and Rat IgG2b κ Isotype control APC-conjugated antibody (553991, BD Bioscience). Samples were tested and analyzed by Longzoe (Shanghai) Biotechnology Co.,
Ltd using BD Fortessa X20 and the FlowJo V10 software, and the company was blinded to the group allocations. LASER SPECKLE Mice were anesthetized by isoflurane, an incision was done along
the midline to separate the skin of the skull, and RFLSI Pro+ laser speckle (RWD Life Science Co., Ltd) was used to detect mice cerebral blood flow. Laser speckle blood flow images were
recorded and used to identify the regions of interest (ROIs). Within these ROIs, the mean blood flow index was calculated in real time. IN VIVO IMAGING Mice were fixed in a stereotactic
frame (RWD) after anesthetization with ketamine hydrochloride, an incision was performed, and the posterior neck muscles were separated to access the cisterna magna. Five µl of visudyne was
injected into the cisterna magna at a speed of 1 µl/min, and the needle was kept in place for 2 min to avoid leakage. Control group mice were not injected with any solution. Fifteen minutes
later, the distribution of visudyne was detected by KODAK In-Vivo Multispectral Imaging System FX using a 630-nm laser for excitation. Then mice were killed to acquire the skulls, and the
visudyne distributions on the skull were also recorded. INDOCYANINE GREEN NEAR-INFRARED (ICG-NIR) IMAGING ICG was dissolved in saline (2 mg/ml, Cat. No. 17478-701-02, Akorn). Mice from the
SAH group (at 7 days post-surgery) and the saline-injected group were fixed in a stereotactic frame (RWD) after anesthetization, and cisterna magna was exposed. Five µl of ICG was injected
into the cisterna magna (1 µl/min), and then the needle was left in place for 2 min to avoid leakage. ICG fluorescence of mandibular LNs and its afferent lymphatics were visualized by an IR
laser (Changchun Laser Technology) and recorded continuously by an Olympus microscope (exposure times 200 ms) for 1 h. The images were analyzed using the Image J software. ROIs were
identified in the afferent lymphatic vessel, and vessel contraction rate (pulse/min) was calculated to present the lymph flow function according to previous studies20,58. TISSUE PROCESSING
dCLNs and mandibular LNs were harvested in a deep anesthesia condition, fixed in 4% paraformaldehyde (PFA) overnight, and then incubated serially in 10%, 20%, and 30% sucrose solutions for 3
days each. For immunofluorescence staining, CLNs were embedded in OCT, and 7-µm-thick sections were sliced by a cryostat (Leica, CM3050S). After transcardial perfusion with saline and 4%
PFA for 15 min, the skullcap was harvested and fixed in 4% PFA overnight, and then the meninges were dissected from the skullcap. IMMUNOFLUORESCENCE For immunofluorescence, the whole mounts
and sections were blocked by 0.3% PBST with 5% bovine serum albumin for 1 h at room temperature, then incubated with primary antibodies overnight at 4 °C. After washing with PBS three times
for 15 min each, secondary antibodies were incubated for 2 h at room temperature. Finally, the whole mounts and sections were mounted with mounting medium with 4,6-diamidino-2-phenylindole
(Cat. No. F6057, Sigma). The primary antibodies used in immunofluorescence included rabbit anti-Lyve-1 antibody (1:1000; Abcam, Cat. No. ab14917), rat anti-Ly76 [Ter119] antibody (1:500;
Abcam, Cat. No. ab91113), rat anti-Ter119 PE-conjugated antibody (1:100; Cat. No. 12-5921-81, eBioscience), rat anti-Lyve-1 eFluor 660-conjugated antibody (1:200; 50-0443-80, eBioscience),
hamster anti-PDPN antibody (1:200; Abcam, Cat. No. ab11936), rabbit anti-Prox1 antibody (1:100; AngioBio, Cat. No. 11-002P), and rat ant-CD31 antibody (Abcam, Cat. No. ab7388). The
corresponding secondary antibodies were used as follows: DylightTM 488-labeled goat anti-rabbit antibody (1:200; KPL, Cat. No. 072-03-15-06), DylightTM 488-labeled goat anti-rat antibody
(1:200; Cat. No. 072-03-16-06, KPL), Alexa Fluor 546 goat anti-hamster antibody (1:200, Invitrogen, Cat. No. A-21111), Alexa Fluor 488/555 goat anti-rat antibody (1:1000, Cell Signaling
Technology, Cat. No. 4416S/4417S), and Alexa Fluor 555 goat anti-rabbit antibody (1:1000, Cell Signaling Technology, Cat. No. 4413S). IMAGE ANALYSIS For the whole-mount staining of meninges,
images were acquired with an Olympus VS120 microscope and a 10× objective with 0.4 NA, or acquired with an Olympus FV1000 confocal microscope and a 40× objective with 0.95 NA, with a
resolution of 1024 × 1024 pixels and a _z_-step of 4 µm. The exposure time and brightness/contrast of each image were applied equally across all images, and images were analyzed using the
Image J (NIH) software. For the CLN sections, images were acquired with an Olympus VS120 microscope and a 20× objective with 0.75 NA. The numbers of erythrocyte per mm2 in the
Lyve-1-positive lymphatic sinus of CLNs were calculated, with only the erythrocytes with intact morphology included. The mean value of five sections of each CLN was used to make a plot
graph. The numbers of erythrocytes in meningeal lymphatics per field were calculated, and four to five fields of each meninges were quantified to acquire the mean value. The percentage of
meningeal lymphatics labeled by AF488 Lyve-1 antibody (i.c.m.) was defined by dividing the area of AF488 Lyve-1 antibody (i.c.m.) labeled by the area of meningeal lymphatics. The percentage
of dCLN lymphatics labeled by AF488 Lyve-1 antibody (i.c.m.) was determined by dividing the area AF488 Lyve-1 antibody (i.c.m.) labeled per section by the area of the dCLN section. The
mircobead coverage in dCLN was quantified by dividing the area of microbeads per section by the area of the dCLN section. Five to ten sections of each dCLN were quantified to acquire the
mean value. Lymphatic ablation and lymphatic regression were measured by dividing the area of Lyve-1 labeled by the area of the sinus, and lymphatic coverage on transverse sinus and sagittal
sinus was calculated separately. Percentage of blood vasculature coverage on sinuses was calculated by dividing the area of the CD31-positive vessels by the area of sinuses. Raw data were
collected using the Microsoft Excel 2007 software. STATISTICAL ANALYSIS Data were expressed as means ± SD, with differences between mean values determined by two-tailed unpaired Student’s
_t_ test, one-way analysis of variance (ANOVA), or two-way ANOVA with Turkey’s multiple-comparison test using the GraphPad Prism 6 Software. _P_ values < 0.05 were considered significant.
The investigators responsible for data analysis were blinded to the group allocations. REPORTING SUMMARY Further information on research design is available in the Nature Research Reporting
Summary linked to this article. DATA AVAILABILITY The raw data underlying Figs. 1c, e, 2b, e, 3b, c, f, 4c–i, 5c, f–l, 6b–e, g, h, j, 7b–e and Supplementary Figs. 1d and 3b, d, h, j are
available via a source data file submitted with this manuscript. All other data are available from the corresponding authors upon reasonable request. REFERENCES * Johnson, C. O. et al.
Global, regional, and national burden of stroke, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. _Lancet Neurol._ 18, 439–458 (2019). Article Google Scholar *
Feigin, V. L. et al. Worldwide stroke incidence and early case fatality reported in 56 population-based studies: a systematic review. _Lancet Neurol._ 8, 355–369 (2009). Article PubMed
Google Scholar * Gijn, J., Kerr, R. S. & Rinkel, G. J. E. Subarachnoid haemorrhage. _Lancet_ 369, 306–318 (2007). Article PubMed Google Scholar * Lieshout, J. H. et al. An
introduction to the pathophysiology of aneurysmal subarachnoid hemorrhage. _Neurosurg. Rev._ 41, 917–930 (2018). Article PubMed Google Scholar * Mees, D. S. M., Bergh, W. M., Algra, A.
& Rinkel, G. J. Antiplatelet therapy for aneurysmal subarachnoid haemorrhage. _Cochrane Database Syst. Rev._ 17, CD006184 (2007). Google Scholar * Etminan, N., Vergouwen, M. D.,
Ilodigwe, D. & Macdonald, R. L. Effect of pharmaceutical treatment on vasospasm, delayed cerebral ischemia, and clinical outcome in patients with aneurysmal subarachnoid hemorrhage: a
systematic review and meta-analysis. _J. Cereb. Blood Flow Metab._ 31, 1443–1451 (2011). Article CAS PubMed PubMed Central Google Scholar * Kasuya, H. et al. Activation of the
coagulation system in the subarachnoid space after subarachnoid haemorrhage serial measurement of fibrinopeptide A and bradykinin of cerebrospinal fluid and plasma in patients with
subarachnoid haemorrhage. _Acta Neuroehir. (Wien)_ 91, 120–125 (1988). Article CAS Google Scholar * Reilly, C., Amidei, C., Tolentino, J., Jahromi, B. S. & Macdonald, R. L. Clot
volume and clearance rate as independent predictors of vasospasm after aneurysmal subarachnoid hemorrhage. _J. Neurosurg._ 101, 255–261 (2004). Article PubMed Google Scholar * Massicotte,
E. M. & Bigio, M. R. D. Human arachnoid villi response to subarachnoid hemorrhage: possible relationship to chronic hydrocephalus. _J. Neurosurg._ 91, 80–84 (1999). Article CAS PubMed
Google Scholar * Nina, P. et al. A study of blood coagulation and fibrinolytic system in spontaneous subarachnoid hemorrhage. _Surg. Neurol._ 55, 197–203 (2001). Article CAS PubMed
Google Scholar * Louveau, A. et al. Structural and functional features of central nervous system lymphatic vessels. _Nature_ 523, 337–341 (2015). Article ADS CAS PubMed PubMed Central
Google Scholar * Aspelund, A. et al. A dural lymphatic vascular system that drains brain interstitial fluid and macromolecules. _J. Exp. Med._ 212, 991–999 (2015). Article CAS PubMed
PubMed Central Google Scholar * Da Mesquita, S. et al. Functional aspects of meningeal lymphatics in ageing and Alzheimer’s disease. _Nature_ 560, 185–191 (2018). Article ADS PubMed
PubMed Central CAS Google Scholar * Louveau, A. et al. CNS lymphatic drainage and neuroinflammation are regulated by meningeal lymphatic vasculature. _Nat. Neurosci._ 21, 1380–1391
(2018). Article CAS PubMed PubMed Central Google Scholar * Wang, L. et al. Deep cervical lymph node ligation aggravates AD-like pathology of APP/PS1 mice. _Brain Pathol._ 29, 176–192
(2019). Article CAS PubMed Google Scholar * Wen, Y. R., Yang, J. H., Wang, X. & Yao, Z. B. Induced dural lymphangiogenesis facilities soluble amyloid-beta clearance from brain in a
transgenic mouse model of Alzheimer’s disease. _Neural Regen. Res._ 13, 709–716 (2018). Article PubMed PubMed Central Google Scholar * Goodman, J. R., Adham, Z. O., Woltjer, R. L., Lund,
A. W. & Iliff, J. J. Characterization of dural sinus-associated lymphatic vasculature in human Alzheimer’s dementia subjects. _Brain Behav. Immun._ 73, 34–40 (2018). Article PubMed
PubMed Central Google Scholar * Hsu, M. et al. Neuroinflammation-induced lymphangiogenesis near the cribriform plate contributes to drainage of CNS-derived antigens and immune cells. _Nat.
Commun._ 10, 229 (2019). Article ADS PubMed PubMed Central CAS Google Scholar * Zhang, Y., Daubel, N., Stritt, S. & Makinen, T. Transient loss of venous integrity during
developmental vascular remodeling leads to red blood cell extravasation and clearance by lymphatic vessels. _Development_ 145, dev156745 (2018). * Ma, Q., Ineichen, B. V., Detmar, M. &
Proulx, S. T. Outflow of cerebrospinal fluid is predominantly through lymphatic vessels and is reduced in aged mice. _Nat. Commun._ 8, 1434 (2017). Article ADS PubMed PubMed Central CAS
Google Scholar * Oliveira Manoel, A. L. & Macdonald, R. L. Neuroinflammation as a target for intervention in subarachnoid hemorrhage. _Front. Neurol._ 9, 292 (2018). Article PubMed
PubMed Central Google Scholar * Li, R. et al. TSG-6 attenuates inflammation-induced brain injury via modulation of microglial polarization in SAH rats through the SOCS3/STAT3 pathway. _J.
Neuroinflammation_ 15, 231 (2018). Article ADS PubMed PubMed Central CAS Google Scholar * Niinimäki, E., Mennander, A. A., Paavonen, T. & Kholová, I. Lymphangiogenesis is increased
in heart valve endocarditis. _Int. J. Cardiol._ 219, 317–321 (2016). Article PubMed Google Scholar * Bouta, E. M. et al. Targeting lymphatic function as a novel therapeutic intervention
for rheumatoid arthritis. _Nat. Rev. Rheumatol._ 14, 94–106 (2018). Article CAS PubMed PubMed Central Google Scholar * Sonia, R., Stephens, M., Roizes, S., Liao, S. & Weid, P. Y.
Acute small intestinal inflammation results in persistent lymphatic alterations. _Am. J. Physiol. Gastrointest. Liver Physiol._ 314, G408–G417 (2018). Article CAS Google Scholar * Becker,
F. et al. A critical role for monocytes_macrophages during intestinal inflammation-associated lymphangiogenesis. _Inflamm. Bowel Dis._ 22, 1326–1345 (2016). Article PubMed PubMed Central
Google Scholar * Zheng, W., Aspelund, A. & Alitalo, K. Lymphangiogenic factors, mechanisms, and applications. _J. Clin. Invest._ 124, 878–887 (2014). Article CAS PubMed PubMed
Central Google Scholar * Wang, X. L., Zhao, J., Qin, L. & Cao, J. L. VEGFR-3 blocking deteriorates inflammation with impaired lymphatic function and different changes in lymphatic
vessels in acute and chronic colitis. _Am. J. Transl. Res._ 8, 827–841 (2016). PubMed PubMed Central Google Scholar * Chen, J. et al. Cerebrovascular injuries induce lymphatic invasion
into brain parenchyma to guide vascular regeneration in zebrafish. _Dev. Cell_ 49, 697.e5–710.e5 (2019). Google Scholar * Yanev, P. et al. Impaired meningeal lymphatic vessel development
worsens stroke outcome. _J. Cereb. Blood Flow Metab._ 40, 263–275 (2020). Article PubMed Google Scholar * Ascenzi, P. et al. Hemoglobin and heme scavenging. _IUBMB Life_ 57, 749–759
(2005). Article CAS PubMed Google Scholar * Garland, P. et al. Heme-hemopexin scavenging is active in the brain and associates with outcome after subarachnoid hemorrhage. _Stroke_ 47,
872–876 (2016). Article CAS PubMed Google Scholar * Pradilla, G., Chaichana, K. L., Hoang, S., Huang, J. & Tamargo, R. J. Inflammation and cerebral vasospasm after subarachnoid
hemorrhage. _Neurosurg. Clin. North Am._ 21, 365–379 (2010). Article Google Scholar * Galea, J. et al. The intrathecal CD163-haptoglobin-hemoglobin scavenging system in subarachnoid
hemorrhage. _J. Neurochem._ 121, 785–792 (2012). Article CAS PubMed PubMed Central Google Scholar * Gallia, G. L. & Tamargo, R. J. Leukocyte-endothelial cell interactions in chronic
vasospasm after subarachnoid hemorrhage. _Neurol. Res._ 28, 750–758 (2006). Article CAS PubMed Google Scholar * Thomas, A. J., Ogilvy, C. S., Griessenauer, C. J. & Hanafy, K. A.
Macrophage CD163 expression in cerebrospinal fluid: association with subarachnoid hemorrhage outcome. _J. Neurosurg._ 131, 47–53 (2018). Article PubMed Google Scholar * Schallner, N. et
al. Microglia regulate blood clearance in subarachnoid hemorrhage by heme oxygenase-1. _J. Clin. Invest._ 125, 2609–2625 (2015). Article PubMed PubMed Central Google Scholar * Xu, Z. et
al. Resident microglia activate before peripheral monocyte infiltration and p75NTR blockade reduces microglial activation and early brain injury after subarachnoid hemorrhage. _ACS Chem.
Neurosci._ 10, 412–423 (2019). Article CAS PubMed Google Scholar * Park, J. H. et al. Cerebrospinal fluid pathways from cisterns to ventricles in N-butyl cyanoacrylate-induced
hydrocephalic rats. _J. Neurosurg. Pediatr._ 8, 640–646 (2011). Article PubMed Google Scholar * Wilson, C. D. et al. Meta-analysis and systematic review of risk factors for shunt
dependency after aneurysmal subarachnoid hemorrhage. _J. Neurosurg._ 126, 586–595 (2017). Article PubMed Google Scholar * Debes, G. F. et al. Chemokine receptor CCR7 required for T
lymphocyte exit from peripheral tissues. _Nat. Immunol._ 6, 889–894 (2005). Article CAS PubMed PubMed Central Google Scholar * Weber, M. et al. Interstitial dendritic cell guidance by
haptotactic chemokine gradients. _Science_ 339, 328–332 (2013). Article ADS CAS PubMed Google Scholar * Lessen, M. et al. Intracellular uptake of macromolecules by brain lymphatic
endothelial cells during zebrafish embryonic development. _Elife_ 6, e25932 (2017). Article PubMed PubMed Central Google Scholar * Breslin, J. W. et al. Lymphatic vessel network
structure and physiology. _Compr. Physiol._ 9, 207–299 (2018). Article PubMed PubMed Central Google Scholar * Dixon, J. B., Raghunathan, S. & Swartz, M. A. A tissue-engineered model
of the intestinal lacteal for evaluating lipid transport by lymphatics. _Biotechnol. Bioeng._ 103, 1224–1235 (2009). Article CAS PubMed PubMed Central Google Scholar * Dixon, J. B.
Mechanisms of chylomicron uptake into lacteals. _Ann. NY Acad. Sci._ 1207, E52–E57 (2010). Article ADS PubMed Google Scholar * Fassbender, K. et al. Inflammatory cytokines in
subarachnoid haemorrhage_ association with abnormal blood flow velocities in basal cerebral arteries. _J. Neurol. Neurosurg. Psychiatry_ 70, 534–537 (2001). Article CAS PubMed PubMed
Central Google Scholar * Atangana, E. et al. Intravascular inflammation triggers intracerebral activated microglia and contributes to secondary brain injury after experimental subarachnoid
hemorrhage (eSAH). _Transl. Stroke Res._ 8, 144–156 (2017). Article CAS PubMed Google Scholar * Schneider, U. C. et al. Microglia inflict delayed brain injury after subarachnoid
hemorrhage. _Acta Neuropathol._ 130, 215–231 (2015). Article PubMed Google Scholar * Zhou, Q., Wood, R., Schwarz, E. M., Wang, Y. J. & Xing, L. Near-infrared lymphatic imaging
demonstrates the dynamics of lymph flow and lymphangiogenesis during the acute versus chronic phases of arthritis in mice. _Arthritis Rheum._ 62, 1881–1889 (2010). PubMed PubMed Central
Google Scholar * Narimatsu, A. et al. Corneal lymphangiogenesis ameliorates corneal inflammation and edema in late stage of bacterial keratitis. _Sci. Rep._ 9, 2984 (2019). Article ADS
PubMed PubMed Central CAS Google Scholar * Wang, X., Zhao, J. & Qin, L. VEGF-C mediated enhancement of lymphatic drainage reduces intestinal inflammation by regulating IL-9_IL-17
balance and improving gut microbiota in experimental chronic colitis. _Am. J. Transl. Res._ 9, 4772–4784 (2017). CAS PubMed PubMed Central Google Scholar * Guo, R. et al. Inhibition of
lymphangiogenesis and lymphatic drainage via vascular endothelial growth factor receptor 3 blockade increases the severity of inflammation in a mouse model of chronic inflammatory arthritis.
_Arthritis Rheum._ 60, 2666–2676 (2009). Article CAS PubMed PubMed Central Google Scholar * Zhou, Q. et al. Vascular endothelial growth factor C attenuates joint damage in chronic
inflammatory arthritis by accelerating local lymphatic drainage in mice. _Arthritis Rheum._ 63, 2318–2328 (2011). Article CAS PubMed PubMed Central Google Scholar * Rahimi, H. et al.
Lymphatic imaging to assess rheumatoid flare: mechanistic insights and biomarker potential. _Arthritis Res. Ther._ 18, 194 (2016). Article PubMed PubMed Central Google Scholar *
Schwager, S. & Detmar, M. Inflammation and lymphatic function. _Front. Immunol._ 10, 308 (2019). Article CAS PubMed PubMed Central Google Scholar * Lin, C. L. et al. A murine model
of subarachnoid hemorrhage-induced cerebral vasospasm. _J. Neurosci. Methods_ 123, 89–97 (2003). Article PubMed Google Scholar * Liang, Q. et al. Lymphatic endothelial cells efferent to
inflamed joints produce iNOS and inhibit lymphatic vessel contraction and drainage in TNF-induced arthritis in mice. _Arthritis Res. Ther._ 18, 62 (2016). Article PubMed PubMed Central
CAS Google Scholar Download references ACKNOWLEDGEMENTS We express gratitude to Fudan University for access to their confocal microscope and their mice behavioral analysis platforms. And
thanks to Dr. Weian Zhang for the in vivo imaging and Professor Beihua Zhang for the laser speckle imaging. We are also grateful to Professor Baohua Tian for his help in the intact
erythrocytes and the degradant differentiation. This work was sponsored by research grants from National Key R&D Program of China (2018YFC1704300 to Y.W.), National Natural Science
Foundation (81822050 and 81920108032 to Q.L., 81904227 to Y.W.), Leading medical talents in Shanghai (2019LJ02 to Q.L.), Dawn plan of Shanghai Municipal Education Commission (19SG39 to
Q.L.), the program for innovative research team of Ministry of Science and Technology of China (2015RA4002 to Y.W.), “Innovation Team” development projects (IRT1270 to Y.W.), Shanghai TCM
Medical Center of Chronic Disease (2017ZZ01010 to Y.W.), Three Years Action to Accelerate the Development of Traditional Chinese Medicine Plan (ZY(2018-2020)-CCCX-3003 to Y.W.), and the
program of Longhua Hospital (KY1932 to Y.W.). AUTHOR INFORMATION AUTHORS AND AFFILIATIONS * Longhua Hospital, Shanghai University of Traditional Chinese Medicine, 725 Wan-Ping South Road,
200032, Shanghai, China Jinman Chen, Hao Xu, Zixin Zhuang, Yangkang Zheng, Xuefei Li, Chinyun Wang, Shaohua Chen, Qianqian Liang & Yongjun Wang * Spine Institute, Shanghai University of
Traditional Chinese Medicine, 725 Wan-Ping South Road, 200032, Shanghai, China Jinman Chen, Hao Xu, Zixin Zhuang, Yangkang Zheng, Xuefei Li, Chinyun Wang, Shaohua Chen, Qianqian Liang &
Yongjun Wang * School of Rehabilitation Science, Shanghai University of Traditional Chinese Medicine, 1200 Cailun Road, 201203, Shanghai, China Jinman Chen, Zixin Zhuang & Yongjun Wang *
Key Laboratory of Theory and Therapy of Muscles and Bones, Ministry of Education (Shanghai University of Traditional Chinese Medicine), 1200 Cailun Road, 201203, Shanghai, China Jinman
Chen, Hao Xu, Zixin Zhuang, Yangkang Zheng, Xuefei Li, Shaohua Chen, Qianqian Liang & Yongjun Wang * Department of Anatomy, School of Basic Medicine, Shanghai University of Traditional
Chinese Medicine, 1200 Cailun Road, 201203, Shanghai, China Linmei Wang * Department of Pathology and Laboratory Medicine and Center for Musculoskeletal Research, University of Rochester
Medical Center, 601 Elmwood Avenue, Rochester, NY, 14642, USA Lianping Xing * The International Education College, Nanjing University of Chinese Medicine, 138 Xianlin Road, 210029, Nanjing,
China Chinyun Wang * The Fourth Clinical Medical College, Guangzhou University of Traditional Chinese Medicine, 232 Huandong Road, 510006, Guangdong, China Zibin Guo Authors * Jinman Chen
View author publications You can also search for this author inPubMed Google Scholar * Linmei Wang View author publications You can also search for this author inPubMed Google Scholar * Hao
Xu View author publications You can also search for this author inPubMed Google Scholar * Lianping Xing View author publications You can also search for this author inPubMed Google Scholar *
Zixin Zhuang View author publications You can also search for this author inPubMed Google Scholar * Yangkang Zheng View author publications You can also search for this author inPubMed
Google Scholar * Xuefei Li View author publications You can also search for this author inPubMed Google Scholar * Chinyun Wang View author publications You can also search for this author
inPubMed Google Scholar * Shaohua Chen View author publications You can also search for this author inPubMed Google Scholar * Zibin Guo View author publications You can also search for this
author inPubMed Google Scholar * Qianqian Liang View author publications You can also search for this author inPubMed Google Scholar * Yongjun Wang View author publications You can also
search for this author inPubMed Google Scholar CONTRIBUTIONS J.C., L.W., H.X., L.X., Q.L., and Y.W. conceived and designed the study; J.C., L.W., S.C., and Z.G. performed the experiments;
Y.Z. performed behavioral tests and data analysis; X.L. analyzed the data of blood flow index and counted the numbers of erythrocytes in CLNs and in meningeal lymphatics; Z.Z. analyzed the
data of lymph flow frequency, counted the numbers of erythrocytes in CLNs, and calculated the percentage of meningeal lymphatics and blood vasculature coverage on sinus; C.W. counted the
numbers of erythrocyte in CLNs and analyzed the area of AF488 Lyve-1 antibody (i.c.m.) labeled lymphatics and mircobead coverage (Y.Z., X.L., Z.Z., and C.W. were blinded to group
allocations); J.C. and Q.L. drafted the manuscript; L.W., H.X., L.X., Q.L., and Y.W. revised the manuscript. All authors have approved the final version of the manuscript and have agreed to
be accountable for all aspects of the work. CORRESPONDING AUTHORS Correspondence to Qianqian Liang or Yongjun Wang. ETHICS DECLARATIONS COMPETING INTERESTS The authors declare no competing
interests. ADDITIONAL INFORMATION PEER REVIEW INFORMATION _Nature Communications_ thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Peer reviewer
reports are available. PUBLISHER’S NOTE Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations. SUPPLEMENTARY INFORMATION
SUPPLEMENTARY INFORMATION REPORTING SUMMARY PEER REVIEW FILE SOURCE DATA SOURCE DATA RIGHTS AND PERMISSIONS OPEN ACCESS This article is licensed under a Creative Commons Attribution 4.0
International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the
source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative
Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by
statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit
http://creativecommons.org/licenses/by/4.0/. Reprints and permissions ABOUT THIS ARTICLE CITE THIS ARTICLE Chen, J., Wang, L., Xu, H. _et al._ Meningeal lymphatics clear erythrocytes that
arise from subarachnoid hemorrhage. _Nat Commun_ 11, 3159 (2020). https://doi.org/10.1038/s41467-020-16851-z Download citation * Received: 22 May 2019 * Accepted: 22 May 2020 * Published: 22
June 2020 * DOI: https://doi.org/10.1038/s41467-020-16851-z SHARE THIS ARTICLE Anyone you share the following link with will be able to read this content: Get shareable link Sorry, a
shareable link is not currently available for this article. Copy to clipboard Provided by the Springer Nature SharedIt content-sharing initiative
Trending News
Access to this page has been deniedYour browser appears to have Javascript disabled.For instructions on how to enable Javascript please click here.If you h...
As digital earth gains momentum, china is setting the paceAl Gore’s 1992 forecast of a Digital Earth — where satellites beam data to reveal all the planet’s environmental dynamic...
Aadhaar-sim linking: faqs to link your mobile number with uidai* Home * Business * Aadhaar-SIM Linking Deadline is February 6, 2018: FAQs to Link Your Mobile Number With UIDAI AT PRES...
Illusions of the Upper Air 1 | NatureABSTRACT A REVIEW OF PROGRESS IN METEOROLOGICAL THEORY IN ENGLAND SINCE 1866. Access through your institution Buy or sub...
“optical glass” and fluorite: an ethical noteABSTRACT PROF. HARTOG, in his “comments” Nos. 2 and 3, revives an old charge which was made by Mr. Lewis Wright in the _...
Latests News
Meningeal lymphatics clear erythrocytes that arise from subarachnoid hemorrhageABSTRACT Extravasated erythrocytes in cerebrospinal fluid (CSF) critically contribute to the pathogenesis of subarachnoi...
Internship program | veterans affairsBRIEFLY DESCRIBE IN NARRATIVE FORM IMPORTANT INFORMATION TO ASSIST POTENTIAL APPLICANTS IN ASSESSING THEIR LIKELY FIT WI...
Body mass index and contralateral ratio predict outcome following unilateral adrenalectomy in primary aldosteronismABSTRACT The effect of unilateral adrenalectomy on blood pressure (BP) outcome in primary aldosteronism (PA) is diverse....
Page Not FoundPage Not Found The content that you're looking for is unavailable. You might find what you are looking for by using the ...
Exclusive-amazon to close all of its physical bookstores and ‘4-star’ shopsExclusive-Amazon to close all of its physical bookstores and ‘4-star’ shops | WTVB | 1590 AM · 95.5 FM | The Voice of Br...