New atpenins, nbri23477 a and b, inhibit the growth of human prostate cancer cells
New atpenins, nbri23477 a and b, inhibit the growth of human prostate cancer cells"
- Select a language for the TTS:
- UK English Female
- UK English Male
- US English Female
- US English Male
- Australian Female
- Australian Male
- Language selected: (auto detect) - EN
Play all audios:
ABSTRACT The growth and metastasis of prostate cancer are regulated by prostate stroma through the tumor–stromal cell interactions. Small molecules that modulate the tumor–stromal cell
interactions will be new anticancer drugs. In the course of our screening of the modulators, we isolated two new atpenins, NBRI23477 A (4) and B (5), from the fermentation broth of
Penicillium atramentosum PF1420. Compounds 4 and 5 as well as atpenin A4 (1), A5 (2) and B (3) inhibited the growth of human prostate cancer DU-145 cells in the coculture with human prostate
stromal cells more strongly than that of DU-145 cells alone. SIMILAR CONTENT BEING VIEWED BY OTHERS NEW DITERPENES FROM THE MARINE SPONGE _SPONGIONELLA_ SP. OVERCOME DRUG RESISTANCE IN
PROSTATE CANCER BY INHIBITION OF P-GLYCOPROTEIN Article Open access 09 August 2022 ANDROPROSTAMINE A: A UNIQUE ANTIPROSTATE CANCER AGENT Article 28 July 2021 NEW CYTOTOXIC DAMMARANE TYPE
SAPONINS FROM _ZIZIPHUS SPINA-CHRISTI_ Article Open access 23 November 2023 INTRODUCTION The growth and metastasis of prostate cancer are regulated by prostate stroma.1, 2 We have reported
earlier that prostate stromal cell (PrSC) promotes the growth of human prostate cancer cells through the secretion of insulin-like growth factor-I.3, 4 There is a possibility that small
molecules could inhibit cancer cell growth by modulating tumor–stromal cell interactions. We developed the _in vitro_ coculture system of human prostate cancer cells and PrSC, in which the
growth of prostate cancer cell is increased by the coculture with PrSC.3, 5 Using the assay method, we have been searching for the modulators of the tumor–stromal cell interactions. In the
course of our screening of the modulators, we isolated new atpenins, NBRI23477 A (4) and B (5), along with the known compounds, atpenin A4 (1), A5 (2) and B (3).6, 7 Here we describe the
isolation, structure determination and biological activity of 4 and 5. We also report the activity of 1, 2 and 3 on our assay. MATERIALS AND METHODS REAGENTS Rhodanile blue was purchased
from Aldrich (Milwaukee, WI, USA). Insulin and hydrocortisone were obtained from Sigma (St Louis, MO, USA). Transferrin was obtained from Wako Pure Chemical Industries (Tokyo, Japan). The
recombinant human basic fibroblast growth factor was purchased from Pepro Tech (London, UK). CELLS The human prostate cancer DU-145 cells were obtained from American Type Culture Collection
(Manassas, VA, USA) and maintained in Dulbecco's modified Eagle's medium supplemented with 10% fetal bovine serum (ICN Biomedicals, Aurora, OH, USA), 100 U ml−1 penicillin G and
100 μg ml−1 streptomycin at 37 °C with 5% CO2. The human normal PrSCs were obtained from Bio Whittaker (Walkersville, MD, USA) and maintained in Dulbecco's modified Eagle's medium
supplemented with 10% fetal bovine serum, 100 U ml−1 penicillin G, 100 μg ml−1 streptomycin, ITH (5 μg ml−1 insulin, 5 μg ml−1 transferrin and 1.4 μM hydrocortisone) and 5 ng ml−1 human
basic fibroblast growth factor at 37 °C with 5% CO2. COCULTURE EXPERIMENT A microplate assay method for the selective measurement of epithelial tumor cells in coculture with stromal cells
using rhodanile blue dye was performed as described before.5 PrSCs were first inoculated into 96-well plates at 5000 cells per well in 100 μl of Dulbecco's modified Eagle's medium
supplemented with ITH and 0.1% fetal bovine serum in the presence of various concentrations of the test compounds. After 2 days, 10 μl of DU-145 cell suspension (5000 cells) in serum-free
Dulbecco's modified Eagle's medium was inoculated onto a monolayer of PrSC, and the cells were further cultured for 3 days. For monoculture of DU-145 cells, the assay medium alone
was first incubated in the presence of test compounds for 2 days at 37 °C. Then, DU-145 cells were inoculated as described above and cultured for further 3 days. ANALYTICAL MEASUREMENT
Melting points were obtained on a Yanagimoto micro melting point apparatus (Yanagimoto, Kyoto, Japan). Optical rotations were measured on a JASCO P-1030 polarimeter (JASCO, Tokyo, Japan). UV
spectra were recorded on a Hitachi 228 A spectrometer (Hitachi, Tokyo, Japan). 1H- and 13C-NMR spectra were measured on a JEOL JNM A400 spectrometer (JEOL, Tokyo, Japan) using TMS as an
internal standard. High resolution electrospray ionization mass spectrometry (HR-ESI-MS) spectra were measured with a JEOL JMS-T100LC spectrometer (JEOL). FERMENTATION OF FUNGAL STRAIN
PF1420 _Penicillium atramentosum_ PF1420 was isolated from a soil sample collected in Iwamizawa, Hokkaido, Japan. A slant culture of _P. atramentosum_ PF1420 was used to inoculate 100-ml
Erlenmeyer flasks. Each contained 20 ml of a seed medium consisting of 2.0% soluble starch, 1.0% glucose, 0.2% soybean meal, 0.6% wheat germ, 0.5% polypeptone, 0.3% yeast extract and 0.2%
CaCO3 in deionized water adjusted to pH 7.2 with NaOH solution before sterilization. The flasks were incubated at 25 °C for 72 h on a rotary shaker at 220 r.p.m. Portions of 1.0 ml of this
seed culture were transferred into six 500-ml Erlenmeyer flasks, each of which contained 100 ml of a seed medium. The flasks were incubated at 25 °C for 48 h on a rotary shaker at 220 r.p.m.
Portions of 150 ml of this seed culture were transferred into four stainless vats, each of which contained 2.5% soybean meal and water-absorbed rice (4 kg) as solid production medium. The
stainless vats were thoroughly stirred and then statically cultured at 25 °C for 14 days. After incubation, 16-kg portion of the obtained culture was extracted with 32 l of 67% aqueous
acetone. RESULTS ISOLATION PROCEDURE FOR ATPENINS The 16-kg culture broth of _P. atramentosum_ PF1420 was extracted with 32 l of 67% aqueous acetone. The filtrate of the extracts was
concentrated _in vacuo_ to remove acetone. The aqueous solution (5 l, pH 7) was applied on an HP-20 column. After washing the column with H2O and 50% MeOH, active ingredients were eluted
with 100% MeOH. The eluate was concentrated _in vacuo,_ dissolved in 600 ml H2O and then extracted with EtOAc. The organic layer was dried over Na2SO4 and concentrated _in vacuo_ to afford
9.92 g of dried material. The materials were applied on a silica gel column (450 g, Wakogel C-200, 75–150 μm; Wako, Osaka, Japan) prepared with CHCl3, and eluted with CHCl3 and CHCl3-MeOH.
The fractions eluted with CHCl3-MeOH (25:1) were concentrated _in vacuo_ to give 4.13 g of viscous material. The viscous material was applied on gel filtration chromatography of Sephadex
LH-20 (MeOH). The fractions containing atpenins were concentrated _in vacuo_ to give 2.76 g of crude material. The crude material was purified by a reversed-phase HPLC column (Inertsil
ODS-3, 20 × 250 mm, 6.0 ml min−1) with 50% CH3CN to afford crude atpenins, 46.4 mg of 1, 119.8 mg of 2, 111.1 mg of 3 and 4, and 14.5 mg of 5. The crude sample of 5 was applied on a Sephadex
LH-20 column (MeOH) to afford pure 8.0 mg of 5. The crude mixture of 3 and 4 was further purified by a reversed-phase HPLC column (Inertsil ODS-3, 10 × 250 mm, 3.0 ml min−1) with 75% CH3CN
and 0.1% TFA, and then the fractions containing 3 and 4 were applied on a Sephadex LH-20 column (MeOH) to afford pure 72.4 mg of 3 and 4.5 mg of 4, respectively. The crude mixture of 1 and 2
was further purified by a reversed-phase HPLC column (Inertsil ODS-3, 10 × 250 mm, 3.0 ml min−1) with 60% CH3CN and 0.1% TFA, and then the fractions containing 1 and 2 were applied on a
Sephadex LH-20 column (MeOH) to afford pure 15.5 mg of 1 and 26.1 mg of 2, respectively. Separation by analytical HPLC (Inertsil ODS-3, 3 μm, 4.6 × 150 mm, 1.0 ml min−1) with 75% CH3CN and
0.1% TFA gave the following retention times (in minutes): 4.38 (1), 4.40 (2), 5.29 (3), 4.95 (4) and 4.28 (5). PHYSICOCHEMICAL PROPERTIES The physicochemical properties of 4 and 5 are
summarized in Table 1. They are soluble in MeOH and DMSO. The molecular formulae of 4 and 5 were determined to be C15H21NO5Cl2 and C15H21NO5, respectively, by HR-ESI-MS. The general features
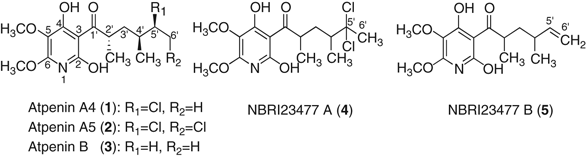
of their UV and NMR spectra resembled each other, indicating structural similarities of these compounds. Compounds 1–3 were identified by NMR spectra as atpenin A4 (1), A5 (2) and B (3),
respectively (Figure 1).6, 7 STRUCTURE DETERMINATION OF NBRI23477 A (4) The 1H- and 13C-NMR data of 4 (Table 2) were similar to those of 2.8 However, the signals of 5′-methine and
6′-methylene of 2 were not observed in the 13C-NMR spectrum of 4, but a quaternary carbon (_δ_ 97.9) and a methyl carbon (_δ_ 35.7) appeared in 4. The partial structure of 4 was established
by analyses of 1H-1H correlation spectroscopy (COSY) and heteronuclear multiple bond connectivity (HMBC) spectra (Figure 2). 1H-1H COSY spectrum revealed the following fragment:
-CH(CH3)-CH2-CH(CH3)-. In the HMBC spectrum, singlet methyl protons (6′-H, _δ_ 2.14) correlated to a quaternary carbon (C-5′, _δ_ 97.9) and a methine carbon (C-4′, _δ_ 48.0). Methyl protons
(_δ_ 1.35) connecting to C-2′ methine correlated to a carbonyl carbon (C-1′, _δ_ 210.6). Therefore, the total structure of 4 was found to be a new family of atpenin (Figure 1). STRUCTURE
DETERMINATION OF NBRI23477 B (5) The 13C- and 1H-NMR data of 5 (Table 2) were similar to those of 3.6, 7 However, the signals of 5′-methylene and 6′-methyl of 3 were not observed in the
13C-NMR spectrum of 5. On the other hand, an olefine carbon (_δ_ 145.2) and a terminal olefine carbon (_δ_ 114.4) were observed in 5. The partial structure of 5 was established by analyses
of the 1H-1H COSY and HMBC spectra (Figure 3). 1H-1H COSY spectrum revealed the following fragment: -CH(CH3)-CH2-CH(CH3)-CH=CH2. In the HMBC spectrum, a methyl proton (_δ_ 1.33) connecting
to C-2′ methine correlated to a carbonyl carbon (C-1′, _δ_ 211.3). Therefore, the total structure of 5 was found to be a new family of atpenin (Figure 1). BIOLOGICAL ACTIVITIES The effects
of 1–5 on coculture of human prostate cancer DU-145 cells with PrSC were determined using rhodanile blue staining method.5 In the coculture, the growth of DU-145 cells is increased by
PrSC.3, 5 As shown in Figure 4, all compounds showed selective growth inhibitory activities and inhibited the growth of DU-145 cells in coculture with PrSC more strongly than that of DU-145
cells alone. The IC50 values of 1–5 against the growth of DU-145 cells in coculture were 0.21, 0.021, 0.034, 0.064 and 0.13 μg ml−1, respectively, whereas those of 1–5 against the growth of
DU-145 cells alone were 0.85, 0.048, 0.54, 0.95 and 0.71 μg ml−1, respectively. All compounds did not show apparent cytotoxicity against stromal cells under microscopic observation (data not
shown). DISCUSSION In this study, we have also obtained three structurally related compounds in addition to 1–5. The HR-ESI-MS spectra revealed that the molecular formulae of these
compounds were C15H22NO6Cl, C15H23NO6 and C15H20NO5Cl3, respectively. Among them, a compound having the molecular formula of C15H20NO5Cl3 may be identical to reported WF-16775 A2,8 but we
could not elucidate the structures of additional three compounds further due to their trace amounts. Ōmura _et al._6 reported that there were atpenins A1, A2 and A3 along with A4, A5 and B,
but they did not obtain A1, A2 and A3 in pure form and did not show any structural information. We cannot exclude the possibility that 4 and 5 would be identical to one of them. However, we
have actually presented here two new structures of atpenins. Atpenins A4, A5 and B were originally isolated as antifungal antibiotics.6, 7 Thereafter, atpenin B was found to decrease the
cellular adenosine 5′-triphosphate.9 Furthermore, it is reported that atpenins specifically inhibit mitochondrial complex II (succinate–ubiquinone oxidoreductase).10 Mitochondria is now
considered as a rational target for cancer therapy.11 Although there is a possibility that atpenins modulate tumor–stromal cell interactions by inhibiting mitochondrial functions, the
elucidation of the precise mechanism of action needs to be studied further. We are now studying the effects of atpenins on tumor growth _in vivo_ using mouse xenograft models. REFERENCES *
Grossfeld, G. D., Hayward, S. W., Tlsty, T. D. & Cunha, G. R. The role of stroma in prostatic carcinogenesis. _Endocr. Relat. Cancer_ 5, 253–270 (1998). Article Google Scholar *
Tuxhorn, J. A., Ayala, G. E. & Rowley, D. R. Reactive stroma in prostate cancer progression. _J. Urol._ 166, 2472–2483 (2001). Article CAS PubMed Google Scholar * Kawada, M., Inoue,
H., Masuda, T. & Ikeda, D. Insulin-like growth factor I secreted from prostate stromal cells mediates tumor–stromal cell interactions of prostate cancer. _Cancer Res._ 66, 4419–4425
(2006). Article CAS PubMed Google Scholar * Kawada, M., Inoue, H., Arakawa, M. & Ikeda, D. Transforming growth factor-β1 modulates tumor–stromal cell interactions of prostate cancer
through insulin-like growth factor-I. _Anticancer Res._ 28, 721–730 (2008). CAS PubMed Google Scholar * Kawada, M. _et al_. A microplate assay for selective measurement of growth of
epithelial tumor cells in direct coculture with stromal cells. _Anticancer Res._ 24, 1561–1568 (2004). CAS PubMed Google Scholar * Ōmura, S. _et al_. Atpenins, new antifungal antibiotics
produced by _Penicillium_ sp. Production, isolation, physico-chemical and biological properties. _J. Antibiot._ 41, 1769–1773 (1988). Article Google Scholar * Kumagai, H., Nishida, H.,
Imamura, N., Tomoda, H. & Ōmura, S. Structures of atpenins A4, A5 and B, new antifungal antibiotics produced by _Penicillium_ sp. _J. Antibiot._ 43, 1553–1558 (1990). Article CAS
Google Scholar * Otsuka, T., Takase, S., Terano, H. & Okuhara, M. New angiogenesis inhibitors, WF-16755A1 and A2. _J. Antibiot._ 45, 1970–1973 (1992). Article CAS Google Scholar *
Oshino, K., Kumagai, H., Tomoda, H. & Ōmura, S. Mechanism of action of atpenin B on Raji cells. _J. Antibiot._ 43, 1064–1068 (1990). Article CAS Google Scholar * Miyadera, H. _et al_.
Atpenins, potent and specific inhibitors of mitochondrial complex II (succinate–ubiquinone oxidoreductase). _Proc. Natl Acad. Sci. USA_ 100, 473–477 (2003). Article CAS PubMed PubMed
Central Google Scholar * Kim, J. & Dang, C. Cancer's molecular sweet tooth and the Warburg effect. _Cancer Res._ 66, 8927–8930 (2006). Article CAS PubMed Google Scholar
Download references ACKNOWLEDGEMENTS We thank Dr R Sawa (Microbial Chemistry Research Center) for analysis of HR-ESI-MS spectra, and Ms K Adachi and Ms E Satoh for their technical
assistance. This work was supported in part by a grant-in-aid from the Ministry of Education, Culture, Sports, Science and Technology of Japan. AUTHOR INFORMATION AUTHORS AND AFFILIATIONS *
Microbial Chemistry Research Center, Numazu Bio-Medical Research Institute, Shizuoka, Japan Manabu Kawada, Isao Momose, Tetsuya Someno & Daishiro Ikeda * Bioscience Labs, Meiji Seika
Kaisha LTD, Kanagawa, Japan Goh Tsujiuchi Authors * Manabu Kawada View author publications You can also search for this author inPubMed Google Scholar * Isao Momose View author publications
You can also search for this author inPubMed Google Scholar * Tetsuya Someno View author publications You can also search for this author inPubMed Google Scholar * Goh Tsujiuchi View author
publications You can also search for this author inPubMed Google Scholar * Daishiro Ikeda View author publications You can also search for this author inPubMed Google Scholar CORRESPONDING
AUTHOR Correspondence to Manabu Kawada. RIGHTS AND PERMISSIONS Reprints and permissions ABOUT THIS ARTICLE CITE THIS ARTICLE Kawada, M., Momose, I., Someno, T. _et al._ New atpenins,
NBRI23477 A and B, inhibit the growth of human prostate cancer cells. _J Antibiot_ 62, 243–246 (2009). https://doi.org/10.1038/ja.2009.20 Download citation * Received: 04 November 2008 *
Revised: 12 February 2009 * Accepted: 23 February 2009 * Published: 13 March 2009 * Issue Date: May 2009 * DOI: https://doi.org/10.1038/ja.2009.20 SHARE THIS ARTICLE Anyone you share the
following link with will be able to read this content: Get shareable link Sorry, a shareable link is not currently available for this article. Copy to clipboard Provided by the Springer
Nature SharedIt content-sharing initiative KEYWORDS * antitumor drug * atpenin * prostate cancer * prostate stroma
Trending News
Vigilance raids eight locations linked to r&b je in da caseNUAPADA: Vigilance officials on Friday conducted simultaneous raids at eight locations linked to junior engineer in the ...
Indian cities: flood of woes, drought of actionUrban development in the Indian context is, effectively, an unplanned amoebic expansion of housing and commercial spaces...
Customer stabs tailor to death over poor trouser fitKANNIYAKUMARI: A restaurant worker was arrested by Vadasery police on Thursday for allegedly stabbing a tailor to death,...
Bjp mp says widows of pahalgam victims 'should have fought back instead of pleading' to terroristsBJP Rajya Sabha MP Ram Chander Jangra on Saturday sparked a controversy after he stated that the widows of the victims o...
Under pressure to hold polls by december, bangladesh's yunus meets bnp, jamaat amid threat to caretaker setupDHAKA: Bangladesh's interim leader, who took over after a mass uprising last year, will meet powerful parties press...
Latests News
New atpenins, nbri23477 a and b, inhibit the growth of human prostate cancer cellsABSTRACT The growth and metastasis of prostate cancer are regulated by prostate stroma through the tumor–stromal cell in...
The page you were looking for doesn't exist.You may have mistyped the address or the page may have moved.By proceeding, you agree to our Terms & Conditions and our ...
The snp’s candidates for the 2021 scottish parliament election — scottish national partyTHE SCOTTISH PARLIAMENT ELECTION ON MAY 6 WILL BE THE MOST IMPORTANT IN SCOTLAND’S HISTORY. Make it Both Votes SNP to re...
Government gets round senate on fuelMotorists will pay more for petrol from November despite the Senate numbers not being there to pass the budget’s fuel ta...
Neurodegeneration studies forge aheadAccess through your institution Buy or subscribe Bing Zhou and colleagues mapped Hallervorden–Spatz syndrome (HSS) — an ...